An Introduction To Flow Batteries
Lithium-ion batteries get all the headlines, but flow batteries are a viable option, particularly for large-scale grid storage.
Lithium-ion batteries have become the energy storage device of choice for cell phones, laptop computers, personal handheld devices, and electric vehicles (EVs). The high energy density of a lithium-ion cell helps it store large amounts of energy without too much weight or taking up too much space. Lithium-ion batteries are also finding use in stationary storage applications such as renewable energy grid storage and backup power supplies for computer systems and critical medical equipment.

Diagram of a flow battery. Image used courtesy of Colintheone, CC BY-SA 4.0, via Wikimedia Commons
Although the price of lithium-ion batteries has come down in recent years, thanks largely to the demand of the EV industry, the technology is still relatively expensive. It has other issues like a limited lifetime and the potential to cause fires if they are over- or under-charged. Traditional lead acid batteries can also be used in these applications but do not have the energy density, charging rate, or capacity that a lithium-ion battery can provide.
Flow Batteries
Lithium-ion batteries are one of many options, particularly for stationary storage systems. Flow batteries store energy in liquid electrolyte (an anolyte and a catholyte) solutions, which are pumped through a cell to produce electricity. Flow batteries have several advantages over conventional batteries, including storing large amounts of energy, fast charging and discharging times, and long cycle life.
The most common types of flow batteries include vanadium redox batteries (VRB), zinc-bromine batteries (ZNBR), and proton exchange membrane (PEM) batteries.
Vanadium Redox
Vanadium redox batteries are the most widely used type of flow battery. They use two different solutions of vanadium ions, one in a positive state (V(+4)) and one in a negative state (V(+5)), which are separated by a membrane. Charging causes the vanadium ions to be oxidized and reduced, causing the electrical potential to increase. When the battery is discharged, the vanadium ions flow through the membrane, generating an electrical current.
Several companies are supplying VRB systems around the world. Invinity Energy Systems has more than 45 megawatt-hours (MWh) of vanadium flow batteries deployed or contracted at sites worldwide. Invinity’s largest installation is a 2 megawatt (MW)/8MWh flow battery co-located with a 6 MW solar photovoltaic (PV) array on land adjacent to Yadlamalka Station, a 1,000 square kilometer sheep and cattle farm in Australia.

Invinity flow batteries are sited at Yadlamalka station in Australia. Image used courtesy of Invinity Energy Systems
Zinc-Bromide
Zinc-bromine (ZNBR) batteries are the oldest type of flow battery (1879) and use zinc and bromine ions to store electrical energy. Their high energy density makes them ideal for large-scale energy storage systems. Zinc-bromine batteries have been used for several decades in various applications, including utility-scale energy storage and backup power systems.
A solution (electrolyte) of zinc-bromine is stored in two tanks. The solution is pumped through a reactor stack comprising two compartments separated by a micro-porous membrane made from polyolefin.
The electrolyte on the negative (anode) side is water-based, while the electrolyte on the positive side contains an organic amine compound. This amine is used to hold the bromine in the solution. When the ZNBR battery is charged, zinc metal is plated as a film on the anode side of a plastic-composite electrode. At the same time, bromide ions oxidize into bromine and form a byproduct that sinks to the bottom of the cathode electrolyte tank. When the battery is discharged, the plated zinc metal is oxidized and dissolved in the aqueous electrolyte, releasing two electrons to the anode used to power an electric device. As the electrons return through the cathode (positive electrode), the bromine in the amine byproduct is converted into two negative bromide ions and combined to form a zinc bromide solution.
Unlike a lithium-ion battery with a 90 percent overall charge-discharge efficiency, a ZNBR is in the 65-75 percent efficiency range. Redflow in Australia and Primus Power in the U.S. are two companies commercializing zinc-bromine flow batteries. A ZNBR battery is scalable through the size of the electrolyte tanks: Redflow makes a 10 kilowatt-hour (kWh) system, while Primus Power has a 125 kWh unit.

ZBM3 zinc bromide flow battery. Image used courtesy of Redflow
Proton Exchange Membrane
Proton exchange membrane (PEM) flow batteries use a proton-conducting membrane to separate the positive (cathode) and negative (anode) electrodes. PEMs are a newer type of flow battery and act as a combination of electrolyzer, using charging electrical energy to split water into hydrogen and oxygen, and a fuel cell, combining the hydrogen and oxygen like a fuel cell during discharge to produce free electrons. A solid-state metal hydride electrode can be used as one of the electrodes to store the hydrogen that forms during charging.
Because of their higher power density, PEM flow batteries are being discussed for use in EVs to replace lithium-ion batteries. Like lithium-ion cells, they exhibit high efficiency, losing little energy between the charge and discharge cycles.
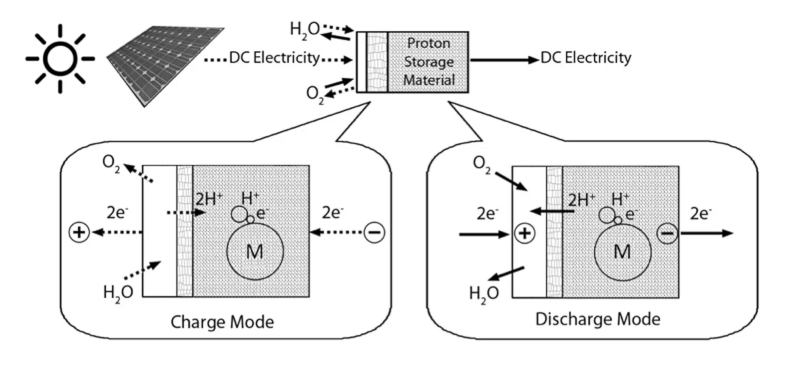
PEM flow battery. Image used courtesy of RMIT University
What’s Next for Flow Batteries?
In the future, flow batteries will play a crucial role in developing renewable energy systems. Renewables like solar and wind energy need energy storage to store excess energy generated during periods of high production and release it during periods of low production. Flow batteries are ideal for this problem, as they can store large amounts of energy and release it quickly when needed.
Flow batteries are also expected to be used in microgrid systems, which are small-scale energy grids independent of the traditional electrical grid. Microgrids are becoming increasingly popular as they provide a more reliable power source, especially in rural areas or areas prone to power outages. Additionally, flow batteries are useful in backup power systems to provide electrical support during power outages and natural disasters.
As the number of EVs on the roads has increased dramatically, the demand for lithium in their batteries will continue to grow. Flow batteries provide a viable alternative to using lithium-ion batteries in grid storage, saving lithium for EVs.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin










