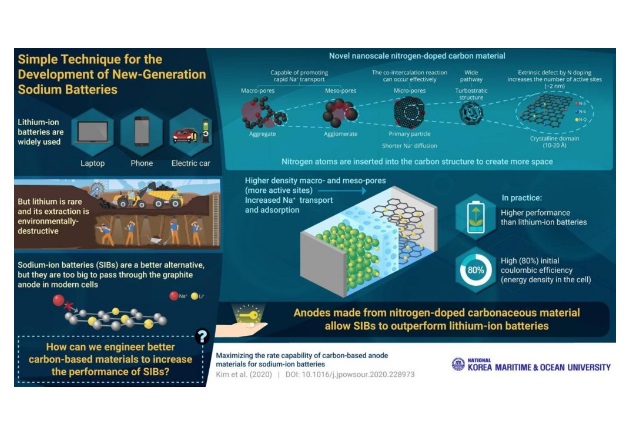
Researchers Look to Improve Rechargeable Sodium-ion Batteries with “Doped” Carbon Anodes
Scientists from the Korea Maritime and Ocean University have published new research focusing on overcoming the limitations of carbon-based anode materials for sodium-ion batteries (SIBs).
This study is the third one of this type by the team, and after phosphorus and sulfur tested in the first two attempts, it focused on the experimentation of “doped” carbon anodes.
Applications for the new technology are varied and include vehicles such as propulsion ships, drones, and more.
Improving the Efficiency of Lithium-ion Batteries
The global demand for lithium-ion batteries is rapidly growing, spearheaded by the increasing number of electric vehicles on the roads.
According to the researchers behind the study, though, lithium is not particularly common or easy to distribute, thus hindering its sustainability while also risking substantial increases in cost for companies.
To circumvent the issue, battery manufacturers have traditionally resorted to SIBs, since they are electrochemically similar to lithium-ion but cheaper.
They also use graphite as a standard anode material, however, which is thermodynamically unstable with sodium ions and can lead to lower reversible capacity and reduced performance.
The Korean research team led by Dr. Jun Kang then started looking for a suitable non-graphite anode material for SIBs.
“Because SIBs have low performance, only 1/10th the capacity of a lithium-ion battery, it is crucial to find an efficient anode that retains graphite’s low cost and stability,” Kang explained.
Experimenting with ‘Doped’ Carbon Anodes
To find such material, the researchers performed a series of tests. First, they explored a hierarchical porous structure capable of promoting rapid sodium transport from the bulk zone of the electrolyte to the interface of the active material.
They then analyzed the retention of large specific surface areas where sodium migrated to the interface, which they found could be easily accessed in the active material.
Next, the team retained the surface defects and pore structures that enable co-intercalation from the surface to the interior, followed by the nanostructures in sodium that were inserted into the active material from defects and pores with short diffusion paths.
Finally, Kang’s team increased the number of active sites due to extrinsic defects that resulted from the elements through hetero-element doping.
Image used courtesy of Korea Maritime and Ocean University.
The process eventually led to improved electrochemical performance of the battery, which was reportedly even superior to traditional lithium-ion batteries.
An Extensive Research
The experiment is the third of this kind, with Kang’s team previously testing the procedure with phosphorus and sulfur, with successful results.
Speaking about potential applications for the technology, the researcher says that most electric vehicles could benefit from it.
Kang directly mentioned electric propulsion ships and other electric vehicles, but also drones, and high-performance CPUs.
More generally, however, the new, five-factor procedure could prove useful in a wide variety of scenarios where sustainable, inexpensive, high-performance batteries are needed.
“These five factors afford good capacity retention, reversible capacity, ultrahigh cycling stability, high initial coulombic efficiency (80%), and remarkable rate capability,” Kang said.
“This means they can be used for a long time even with intense battery use,” he concluded.
For more information about the new study, you can read it in its entirety here.
What do you think about it? Let us know in the comments down below.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin