Researchers Pinpoint Ideal Salt Concentration for Water-Based Zinc Batteries
A new study unveils the ideal electrolyte solution to ensure the greatest balance of performance and reliability.
As the demand for higher performance and more reliable batteries continues to increase, many are exploring solutions outside lithium-ion batteries.
Packed lithium battery with BMS. Image used courtesy of Adobe Stock
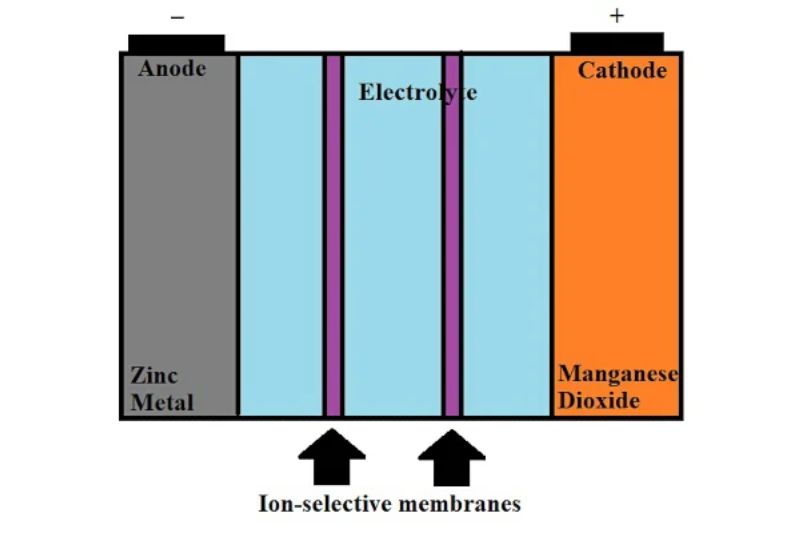
Specifically, one battery chemistry that is gaining interest from researchers is water-based zinc-metal batteries. In these batteries, zinc metal is used as the anode, while an aqueous solution is used as the electrolyte.
Despite the potential for this battery chemistry, several significant engineering challenges still need to be overcome before their potential can be fulfilled. Researchers have recently made significant strides toward commercializing these zinc-based batteries.
In this piece, we’ll look at the advantages of zinc batteries, outstanding engineering challenges, and the new research from ETH Zurich.
Zinc Battery Advantages
There are a number of reasons why water-based zinc-metal batteries are gaining interest as a potential alternative to lithium-ion batteries.
One major reason is that the material properties of zinc enable zinc-metal-anode batteries with extremely high theoretical gravimetric and volumetric capacities of ∼820 mAh g−1 and ∼5800 mAh cm−3, respectively. These high capacities directly translate into a higher theoretical storage capability of zinc batteries. According to certain studies, zinc-based batteries are believed to be ten times better than lithium-ion batteries of the same volume.
The construction of a zinc-ion battery. Image used courtesy of Chetan Kumbhar
Beyond the improvement in capacity that comes from zinc batteries, another major benefit of the technology is its sustainability. Lithium is a depleting resource that also requires extremely destructive mining processes to obtain from the earth. Zinc, on the other hand, is extremely abundant on Earth. It is estimated that zinc consumes 1.9 billion tons of natural resources worldwide and is roughly ⅓ the cost of lithium.
Zinc Battery Challenges
Although zinc offers many potential benefits compared to lithium, significant engineering challenges must be overcome before commercialization.
One significant challenge is that when zinc batteries are charged to high enough voltages, the water in the electrolyte solution undergoes a chemical reaction at the electrode that produces hydrogen gas. When this reaction occurs, the zinc batteries experience an increase in pressure that is dangerous for battery operation, decreasing the reliability of these batteries.
Another impact of this reaction is that the electrolyte loses water content. This results in a more viscous electrolyte solution, which leads to impactfully slower charging and discharging times for the battery.
ETH Research
Recently, researchers from ETH Zurich have made some headway toward improving zinc-ion battery performance and reliability.
As their recently published paper described, the researchers sought to determine the ideal electrolyte solution (i.e., water-to-salt ratio) for minimizing unwanted reactions in the electrolyte. Their experiments used sophisticated computer programs for X-ray absorption spectroscopy (XAS) and molecular dynamics (MD) to simulate the performance of different ratios and observe the resulting performance.
XAS analysis of electrolyte solutions as a function of water-to-cation ratio. Image from Vazquez et al.
Surprisingly, the major study finding was that the best performance was achieved when the water-to-cation ratio was 10 to 5. Before these results, researchers believed that, for the best performance, the aqueous solution should have the highest water-to-salt ratio possible.
With this ratio, the researchers observed a zinc-based battery that minimizes electrolyte depletion and prevents dendrites from forming. The resulting battery simultaneously offered a coloumbic efficiency greater than 99%, a high rate, and greater reliability.
According to the paper, the researchers intend to continue this study by scaling their testing to see the experimental results in larger batteries. With their new finds, the researchers are optimistic that they have paved a path toward commercializing zinc-metal water-based batteries.