Cornell Researchers Use X-Rays to Unveil Transient Defects in Sodium-Ion Batteries
A new study from Cornell hopes to provide greater insight into battery design and performance. This article examines the intercalation process for sodium-ion batteries, challenges with dislocation, and the new research from Cornell.
One of the greatest challenges researchers in the battery industry aim to solve is how to create more sustainable batteries than lithium-ion solutions. The issue is that lithium itself is not particularly abundant and has an environmentally-hazardous supply chain.
Battery research. Image used courtesy of Adobe Stock
Researchers are turning to other battery chemistries like sodium-ion built from more abundant and sustainable materials to solve this issue. Researchers from Cornell University are using advanced imaging techniques to unveil new details of the behavior of sodium-ion batteries.
Sodium-Ion Intercalation
To better understand the research from Cornell, we first must discuss intercalation in sodium-ion batteries.
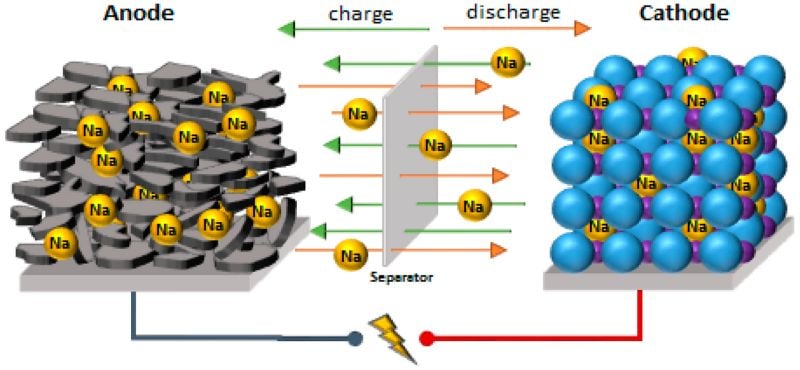
Intercalation is a fundamental process in sodium-ion batteries. Image used courtesy of Peters et al.
In sodium-ion batteries (and lithium-ion batteries), intercalation is the process in which ions are inserted into and removed from layers in a material to facilitate the storage and release of electrical energy. During the discharging process in a sodium-ion battery, Na+ ions are released from the anode, diffuse through the electrolyte solution, and intercalate into the cathode. To compensate for this movement of positively charged ions into the cathode, electrons also move from the anode toward the cathode via an external circuit. It is this process which allows these batteries to source electrical power. During the charging cycle, the opposite happens.
Hence, intercalation is a crucial process for sodium-ion batteries, as the process is responsible for the charging and discharging of the battery.
Dislocation Challenges
While intercalation is a fundamental process in sodium-ion batteries, some challenges occur. Specifically, the challenge of transient dislocation causes cathode degradation in these batteries.
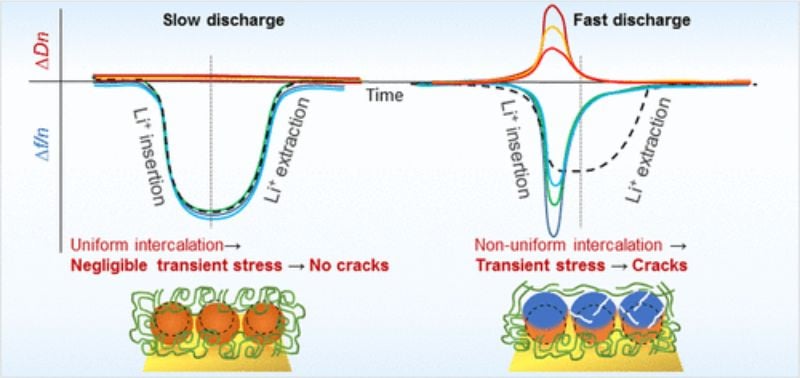
Transient dislocation in lithium battery electrodes. Image used courtesy of Shpigel et al.
When sodium ions are inserted or extracted from the intercalation host material during battery charging or discharging, it can cause changes in the structure of the host material. Specifically, the movement of the ions can lead to temporary defects or distortions in the atomic arrangement, referred to as "transient dislocations." These dislocations can occur as the ions try to navigate through the material, causing the atomic layers to shift or slip past one another.
These dislocations are called “transient” because materials exhibit “self-healing” properties, restoring the material to its original orientation after the charging or discharging. However, while the materials can self-heal, they are still left with “scars” from the process, which eventually build up over time and cause degradation to the cathode material.
Cornell Research
Recently, researchers from Cornell published a paper in which they sought to better understand the phenomenon of transient dislocations due to the intercalation process in sodium-ion batteries.
In the study, researchers used Bragg Coherent Diffractive Imaging with a synchronized X-ray beam to perform imaging of the sodium-ion battery during the charging process. With this specialized imaging technique, the team could create 3D renderings of the battery during the charging process, allowing them to image the transient desolation of the cathode.
The team found the transient dislocations occurred at a specific location, called the anti-phase domain boundary. In this region, a crystal's atoms are configured opposite in a lattice system.
Thanks to this research, the Cornell team has gained greater insight into the physical mechanisms of transient dislocations. It will launch a new study to understand how these dislocations interact with the intercalation process.