Research Advances Hope for Scalable Green Hydrogen
Green hydrogen is emerging as a promising fuel alternative for carbon-free combustion. The options for green hydrogen production are growing, but finding cost-effective solutions at scale is difficult.
The global energy landscape is shifting toward sustainability, driven by the urgent desire to mitigate climate change and reduce dependency on fossil fuels. A promising fuel alternative emerging in this transition is hydrogen, which produces water during combustion and is thus considered a nonpolluting fuel.
Unfortunately, the most common hydrogen production methods rely on natural gas or coal, which generate substantial carbon dioxide emissions, negating any potential environmental advantages associated with hydrogen utilization.
The good news is that even hydrogen has a clean alternative: green hydrogen.
Green hydrogen using renewable energy sources. Image used courtesy of Adobe Stock
Green hydrogen production uses electrolysis powered by renewable sources such as wind and solar energy. Its use in various sectors, including transportation, industry, and heating, holds immense potential for decarbonization.
The challenge for green hydrogen is producing at scale while remaining cost-effective. Understanding the benefits of green hydrogen in the long term, several groups have started to develop technologies to ease the financial challenges of production.
Changing the Catalyst in Green Hydrogen Production
A partnership between the Ulsan National Institute of Science and Technology (UNIST) and the Korea Advanced Institute of Science and Technology (KAIST) may facilitate green hydrogen generation in ecologically sustainable and financially viable ways.
The collaborative endeavor has developed a bifunctional water electrolysis catalyst, supplanting costly precious metal catalysts. This catalyst ensures both high efficiency and stability, enabling the production of high-purity green hydrogen at a remarkable level.
By harnessing the properties of ruthenium, silicon, and tungsten, this catalyst offers a more economical solution compared to traditional platinum or iridium catalysts. It substantially reduces greenhouse gas emissions, positioning it as a sustainable and environmentally friendly alternative.
Using an Aqueous Electrolyte in Hydrogen Electrolysis
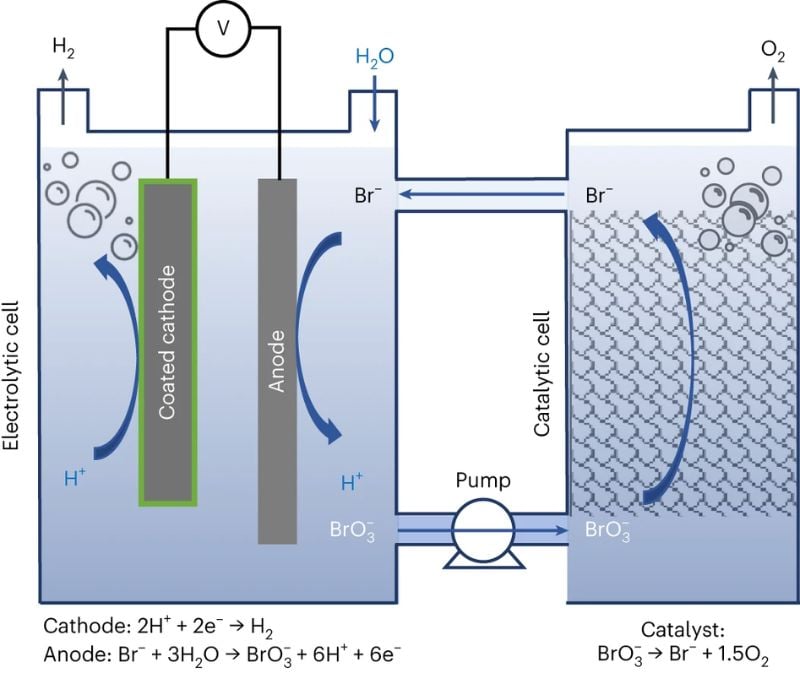
A research team from Technion Israel Institute of Technology previously introduced an efficient electrolysis method called E-TAC, which produced hydrogen and oxygen in the same cell but at different stages. The team has now developed an innovative method of generating both gases simultaneously but within two separate cells.
This approach overcomes operational challenges and constraints associated with the solid electrode used in the E-TAC technique, where oxygen production occurs. Instead, it substitutes the solid electrode with sodium bromide aqueous electrolyte in water, which enables a continuous process and eliminates the need to alternate cold and hot electrolytes through the cell.
Illustration of the decoupled water electrolysis process. Image used courtesy of Nature
The bromide anions within the electrolyte undergo oxidation to bromate, while hydrogen is produced at the cathode. Subsequently, these ions and the aqueous electrolyte flow to a separate cell, where they revert to their original state, simultaneously generating oxygen. This cycle continues continuously, facilitating the production of both gases.
The research showcases the proposed process’s initial feasibility and illustrates its efficiency and capability to operate at high electric currents. This capability indicates that hydrogen production can be achieved at an elevated rate.
Improving Solar-Powered Hydrogen Production
Another team from UNIST is examining how to address the challenges of photoelectrodes in the electrolysis process.
This breakthrough is distinguished by its success in overcoming the limitations of perovskite solar cells (PSCs) and enlarging the photoelectrodes 10,000 times.
The team used perovskite as the photoelectrode material. Enlarging the photoelectrodes allowed this process to overcome efficiency issues with PSCs and increase the efficiency, durability, and scalability of using solar energy for green hydrogen production.
The team additionally opted for formamide instead of methylammonium as the cation in perovskite, a switch that notably bolstered the stability of the photoelectrodes against ultraviolet rays. The contact surface of the photoelectrodes is secured with water using a nickel foil, guaranteeing stability even when wet.
In this configuration, the module has achieved a solar hydrogen conversion efficiency of over 10 percent, meeting the criteria for commercial viability and securing the world’s highest efficiency record for large-area photoelectrodes.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin