High-Voltage Rechargeable Aqueous Battery Aims to Challenge Li-ion Dominance
A new rechargeable high-voltage manganese dioxide zinc battery, exceeding the 2V barrier in aqueous zinc chemistry, is the latest invention by City College of New York researchers. With a voltage of 2.45- to 2.8-V, the alkaline MnO2|Zn battery, developed by Dr. Gautam G. Yadav and his group in the CCNY-based CUNY Energy Institute, could break the long dominance of flammable and expensive lithium (Li)-ion batteries in the market.
To break the previously daunting 2V barrier in aqueous zinc chemistry, primary inventor Yadav and his team interfacially engineered two different aqueous electrolytes that deliver the theoretical capacity (308mAh/g) reversibly for many cycles.
"The voltage of current commercially available alkaline MnO2|Zn batteries is around 1.2- to 1.3-V, and this has been considered low compared to Li-ion which has a voltage greater than 3V," said Yadav.
Voltage has been Li-ion's greatest asset and has helped fuel its rise in an energy hungry world.
"Unfortunately it contains elements that are toxic and geopolitically sensitive with Asian countries having a monopoly on mining and manufacturing them," added Yadav.
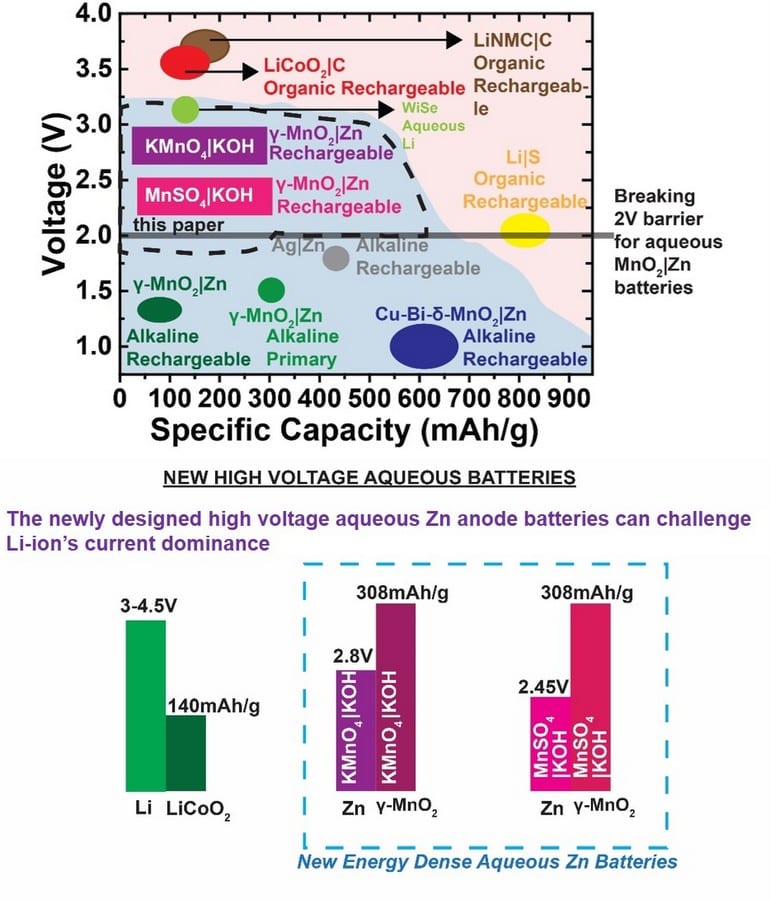 (click on image to enlarge) Photo credit: G.G. Yadav et al, ACS Energy Lett., 2019, 4, 2144-2146.
(click on image to enlarge) Photo credit: G.G. Yadav et al, ACS Energy Lett., 2019, 4, 2144-2146.
"This has put the United States at a tremendous disadvantage and has lost its lead in energy storage industry, when in the past it was a world leader. With manganese and zinc being widely available elements, and with the U.S. being rich with them as well, it allows the U.S. to compete again. The manufacturing cost of these batteries will also be low, so it can kick start the growth of the energy storage industry in the U.S."
Yadav's collaborators from CCNY's Grove School of Engineering included: Xia Wei, Jinchao Huang, Damon E. Turney and Sanjoy Banerjee, who is Distinguished Professor of Chemical Engineering and director of the CUNY Energy Institute.
Their research appears in the journal ACS Energy Letters, which publishes breakthrough energy-related research.