PTO Cathode Enhances Stability and Energy Density of Li-Ion Batteries
Solid-state sodium-ion batteries are known to be much safer than traditional lithium-ion batteries, which have a risk of fire and explosions. However, so far, their performance has not been sufficient to offset the safety advantages.
Researchers from the University of Houston reported the development of an organic cathode that drastically enhances both stability and energy density.
The better performance, which they reported in the journal Joule, is related to two key findings.
First, they found that the resistive interface between the electrolyte and cathode that usually forms during cycling can be reversed, extending cycle life
Secondly, they revealed that the flexibility of the organic cathode let it maintain contact at the interface with the solid electrolyte, even while the cathode expanded and contracted during cycling.
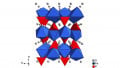
Yan Yao, associate professor of electrical and computer engineering at the University of Houston and co-author of the paper, said the organic cathode that is called PTO (for pyrene-4,5,9,10-tetraone, see molecular model above in which red spheres represent oxygen atoms and gray spheres represent hydrogen atoms) offers unique advantages over previous inorganic cathodes. Yan Yao also noted that the underlying principles are equally significant.
"We found for the first time that the resistive interface that forms between the cathode and the electrolyte can be reversed," Yao said. "That can contribute to stability and longer cycle life." Yao also serves as principal investigator at the Texas Center for Superconductivity at UH. His research group focuses on green and sustainable organic materials for energy generation and storage.
Yanliang "Leonard" Liang, a research assistant professor in the UH Department of Electrical and Computer Engineering, indicated that reversibility of the interface is the key, thereby allowing the solid-state battery to achieve a higher energy density without sacrificing cycle life.
Ordinarily, a solid-state battery's ability to store energy stops when the resistive cathode-electrolyte interface forms. So, reversing that resistance allows energy density to remain high during cycling, Leonard said.
Lithium-ion batteries with their liquid electrolytes can store relatively high amounts of energy and are frequently used to power the tools of modern life. However, the risk of fire and even explosions has intensified interest in other types of batteries, and a solid-state sodium-ion battery promises increased safety at a lower cost.
Xiaowei Chi, a post-doctoral researcher in Yao's group, pointed out that a primary challenge had been finding a solid electrolyte that is as conductive as the liquid electrolytes in lithium-ion batteries. Now that sufficiently conductive solid electrolytes are available, another challenge has been the solid interfaces.
One issue with a solid electrolyte is that the electrolyte has to maintain intimate contact with a traditional rigid cathode even during battery cycling as the cathode expands and contracts. Fang Hao, a PhD. student working in Yao's group, noted that the organic cathode is more pliable and thus can remain in contact with the interface, improving cycling life.
The researchers found that the contact remained steady through at least 200 cycles.
"If you have reliable contact between the electrode and electrolyte, you will have a great chance of creating a high-performance solid-state battery," Hao said.
In addition to Yao, authors of the include co-first authors Hao and Chi, Liang, Ye Zhang and Hui Dong, all of UH; Rong Xu and Kejie Zhao of Purdue University; and Hua Guo, as well as Tanguy Terlier and Jun Lou of Rice University. The majority of this work was funded via the U.S. Department of Energy Advanced Research Projects Agency-Energy.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin