Sodium-aluminum Battery Heats Up Grid Storage
A new design of a molten sodium battery could find large-scale grid storage applications.
Intermittent renewable energy sources are only practical for large-scale power grid integration if paired with energy storage. The capability of storing energy when renewable resources are abundant and releasing those resources at night or when demand is high is one factor driving advances in battery technology.
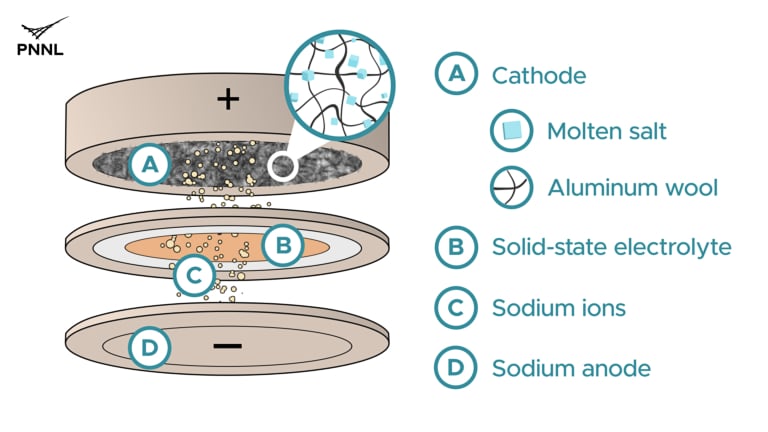
Diagram by Sara Levine, Pacific Northwest National Laboratory
In addition to their primary role in electric vehicles (EVs), cell phones, and personal electronics, Lithium-ion batteries have become the battery design of choice for short-term (less than ten hours) energy storage. Meeting the need for longer-term storage will require a switch to different battery storage technologies.
Molten Sodium
One technology that has been under investigation since the 1980s uses a metal-based cathode (positive electrode) and a molten sodium anode (negative electrode) enclosed within a steel case. A ceramic isolator allows ions to pass between the electrodes, but not electrons, with travel through an external circuit during charging and discharging. Because the sodium must be kept in a molten state, these batteries must operate at temperatures above 250°C, making them impractical for use in EVs but acceptable for stationary grid storage applications.
Recently, a research team from the Department of Energy’s Pacific Northwest National Laboratory (PNNL) announced that they had created a new battery design that could be used for grid energy storage and is particularly suited to 12-24 hour energy storage periods. The PNNL battery uses low-cost and Earth-abundant sodium and aluminum in a design that operates at lower temperatures than conventional high-temperature sodium batteries. The battery, which maintains excellent energy storage capacity, can also charge and discharge much faster than previous molten sodium designs.

Renewable energy storage. Image used courtesy of Adobe Stock
Solid-state Electrolyte
In developing the new battery, PNNL scientists collaborated with U.S.-based Nexceris, applying that company’s solid-state, sodium-based electrolyte to allow sodium ions to travel from the anode to the cathode as the battery charges.
The design uses a flat configuration rather than a more traditional tubular shape. This will allow the individual cells to be stacked to build grid-scale battery capacity. The cathode comprises aluminum wool (a scarp product from aluminum manufacturing) immersed in molten sodium salts. Only sodium ions can pass through the solid electrolyte to reach the sodium metal anode. Cell capacity can be increased by increasing the thickness of the cathode.
During the development of the battery chemistry, the team discovered that changing the neutral molten salt into an acidic molten salt increased the battery capacity. After 345 charges and discharge cycles, the acidic cell retained 82.8 percent of its peak charge capacity.
Adequate Energy Density
Typically, a commercial lithium-ion battery has a specific energy charge density of 170-250 watt-hours per kilogram (Wh/kg) while the PNNL sodium-aluminum battery has delivered an energy density of around 100 Wh/kg. As the PNNL design is destined for stationary applications and is made from much more abundant and inexpensive materials easily available in the U.S., the design is viewed as having potential for grid storage applications.
The PNNL battery also operates at temperatures as much as 100°C lower than commercially available sodium batteries, which means that it can be manufactured with less expensive materials instead of the complex components required for higher temperature designs.
As with any new battery technology, this one is years away from commercial application. However, the initial work looks promising and, with more development, could eventually find its way into grid-scale energy storage.






