Supercrystal Catalyst Enables Efficient Hydrogen Generation From Solar
Researchers have developed a photocatalyst that efficiently produces hydrogen upon solar irradiance.
Photovoltaic technology has advanced remarkably in recent years with evolving material science and manufacturing processes. In seeking higher efficiency and lower costs, researchers have explored novel materials beyond silicon, such as perovskites, graphene, and bio-based substances. Inventions such as multijunction cells can absorb different wavelengths of light, increasing the theoretical efficiency of photovoltaic cells. Traditional solar panel manufacturing methods, such as wafering and chemical etching, have also improved to enhance efficiency.
Illustration of the new 2D super crystal as a photocatalyst. Image used courtesy of LMU
A team at Ludwig Maximilian University's (LMU) Nano-Institute, led by Professor Emiliano Cortés, is transforming solar energy capture through nanostructures. They are developing plasmonic nanostructures that produce green hydrogen from formic acid from sunlight.
Capturing Solar Energy Efficiently
Solar panels compensate for their low power conversion efficiency by covering large areas to capture more sunlight. The LMU researchers’ approach relies on improving the energy capture per area. Their plasmonic nanostructures can concentrate solar energy.
Cortés and Dr. Matías Herran from Fritz Haber Institute, Berlin, along with partners from the Free University of Berlin and the University of Hamburg, demonstrate a two-dimensional supercrystal able to produce hydrogen with sunlight. The new material can function as a photocatalyst with hydrogen as an energy carrier.
The supercrystal offers several advantages over traditional photocatalysts. The ordered arrangement of nanoparticles creates a more efficient light-harvesting system, while the reduced dimension enhances charge transport and separation, further boosting the overall reaction rate. Also, when hydrogen is produced through photocatalysis, it has the potential to serve as a renewable energy carrier, complementing solar energy. It can be stored and transported for various applications, including fuel cells, transportation, and heating.
The researchers create particles in the range of 10-200 nanometers from gold for their supercrystal. Visible light interacts strongly with metal electrons, causing them to oscillate. These oscillations create a dipole moment, allowing them to capture more sunlight and convert them into high-energy electrons. The researchers describe their crystal as a superlens concentrating energy.
Localized High-Energy Hotspots Accelerate Hydrogen Production
The material consists of gold nanoparticles embedded close to each other without contact for maximum light-matter interaction. The researchers noticed that the light absorption increased many times with this arrangement. This enhancement is due to localized strong electric fields called hotspots.
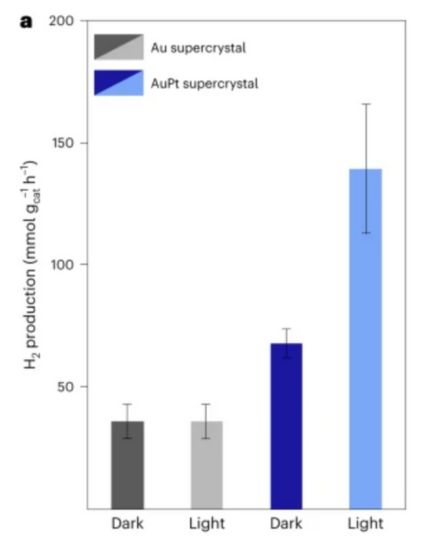
To harness the full potential of produced formic acid, the researchers at LMU teamed up with researchers at the University of Hamburg. They placed platinum in the interspaces between gold particles to improve absorption and accelerate the redox reaction involved in hydrogen production. The reaction converts formic acid into hydrogen. The supercrystal catalyst (per gram) currently produces 139 millimoles of hydrogen from formic acid per hour.
Hydrogen production of supercrystals. Image used courtesy of LMU
Researchers worldwide are focusing on new technologies that use alternative feedstocks for hydrogen fuel, like formic acid, ammonia, and water. The LMU researchers believe their photocatalysts can be a potential solution for realizing this next-generation technology.